Electrons | Vibepedia
Electrons are fundamental subatomic particles carrying a negative elementary electric charge. Their movement is the basis for electricity, powering everything…
Contents
Overview
The concept of the electron as a discrete particle emerged in the late 19th century, a period of intense scientific inquiry into the nature of electricity and matter. While early experiments with cathode rays by scientists like William Crookes hinted at negatively charged entities, it was J.J. Thomson at the Cavendish Laboratory in Cambridge who, in 1897, definitively identified the electron. Thomson's experiments with cathode ray tubes demonstrated that these rays were composed of particles much lighter than atoms, which he initially called 'corpuscles.' His work, building on the understanding of electromagnetism and atomic structure, established the electron as a fundamental constituent of matter, challenging the prevailing view of atoms as indivisible spheres. This discovery was a pivotal moment, opening the door to the field of particle physics and the development of atomic models by figures like Ernest Rutherford and Niels Bohr.
⚙️ How It Works
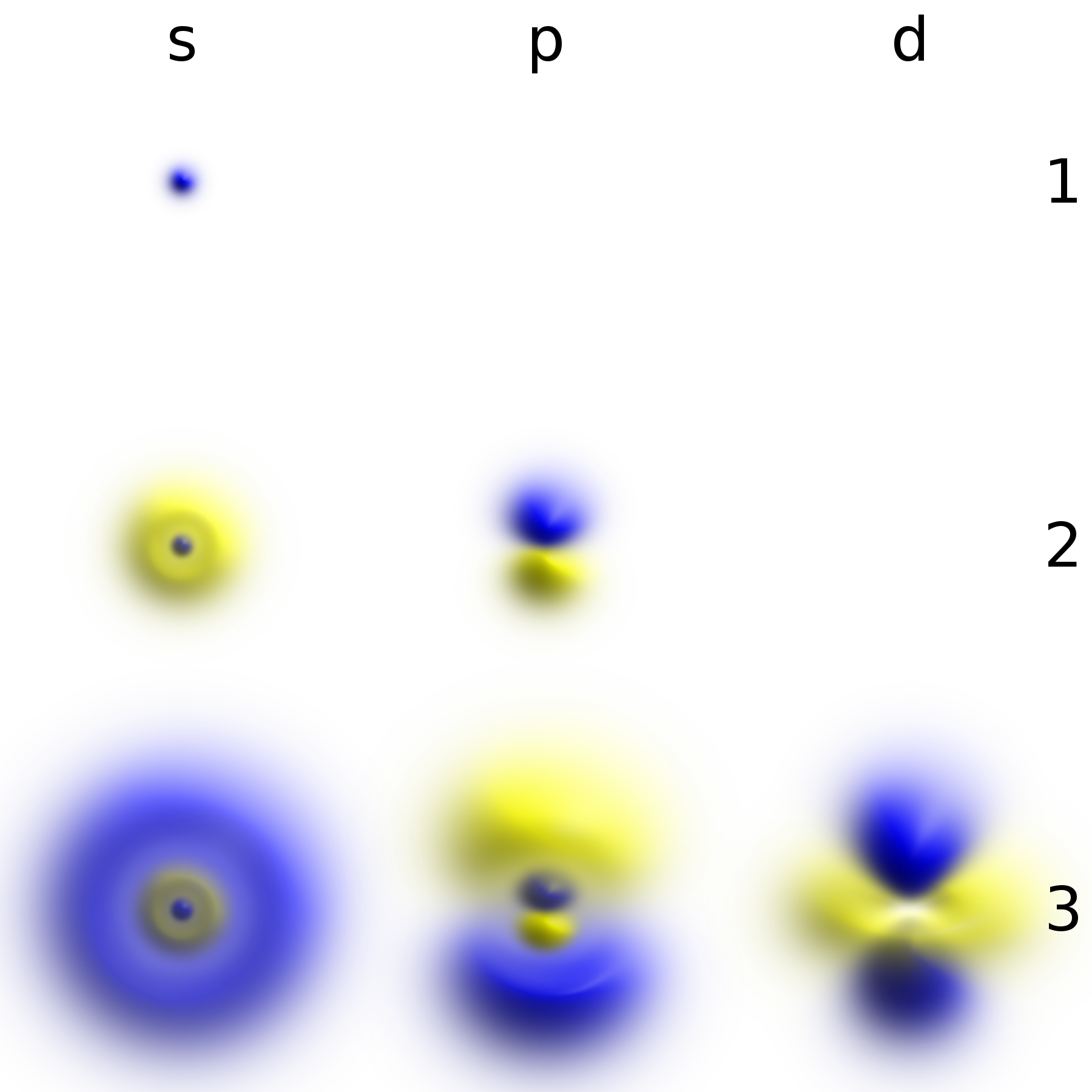
Electrons function as the primary carriers of negative charge in the universe. In an atom, they occupy distinct energy shells or orbitals around the positively charged nucleus, governed by quantum mechanical principles. The arrangement and energy levels of these electrons determine an atom's chemical properties. Specifically, the valence electrons—those in the outermost shell—are the most mobile and are responsible for forming chemical bonds through sharing or transfer, leading to the creation of molecules and complex structures. The flow of these mobile electrons is the essence of electric current, enabling phenomena from static electricity to the operation of electronic devices. Their wave-particle duality means they can exhibit characteristics of both waves and particles, a cornerstone of quantum mechanics.
📊 Key Facts & Numbers
An electron possesses a mass of approximately 9.109 x 10^-31 kilograms, making it about 1/1836th the mass of a proton. Its electric charge is precisely -1.602 x 10^-19 coulombs, defined as one elementary charge. In a typical atom, such as carbon-12, there are six electrons. A single copper wire can contain an estimated 10^23 free electrons per cubic centimeter. The flow of electrons in a circuit is measured in amperes, where one ampere equals approximately 6.24 x 10^18 electrons passing a point per second. The energy levels of electrons in atoms are quantized, with the smallest energy difference between levels in hydrogen being about 1.6 x 10^-18 joules, corresponding to the energy of a photon of ultraviolet light.
👥 Key People & Organizations
Key figures in the electron's story include J.J. Thomson, credited with its discovery in 1897, and Robert Millikan, who precisely measured the electron's charge in his famous oil-drop experiment around 1909. G.P. Thomson (son of J.J.) provided experimental evidence for the wave nature of electrons in 1927, a discovery that earned him the Nobel Prize in Physics. Organizations like the Cavendish Laboratory at the University of Cambridge and Bell Laboratories have been central to electron-related research, from early discoveries to the development of semiconductor devices. The American Physical Society and the European Physical Society continue to foster research in this fundamental area.
🌍 Cultural Impact & Influence
The electron's influence permeates nearly every facet of modern existence. Its discovery fundamentally altered our understanding of matter and energy, leading directly to the development of atomic theory and quantum mechanics. The ability to control and manipulate electron flow underpins the entire field of electronics, from the vacuum tubes of early radios to the microprocessors in today's computers and smartphones. Chemical bonds, formed by electron interactions, are the basis of all biological processes and material science, from the DNA in our cells to the plastics in everyday objects. The electron's role in light emission and absorption is also fundamental to technologies like LEDs and lasers.
⚡ Current State & Latest Developments
Current research continues to explore the exotic behaviors of electrons, particularly in condensed matter physics. Phenomena like superconductivity, where electrons flow with zero resistance, and quantum entanglement involving electrons, are areas of intense study. The development of quantum computing relies heavily on manipulating electron states, such as their spin, to perform complex calculations. Advances in nanotechnology allow for the precise control of individual electrons, opening doors to novel electronic devices and sensors. The ongoing quest to unify quantum mechanics with general relativity also involves understanding the electron's place within a more complete theory of fundamental forces.
🤔 Controversies & Debates
One persistent debate revolves around the precise nature of the electron's wave-particle duality. While quantum mechanics provides an incredibly accurate predictive framework, the underlying 'reality' of what an electron is remains a subject of philosophical discussion. Another area of contention, particularly in the context of quantum field theory, is the concept of the electron's 'bare' charge versus its 'dressed' charge, which is influenced by quantum fluctuations. Furthermore, the search for supersymmetric particles, which would imply a partner particle for the electron, remains a significant, albeit unconfirmed, pursuit in high-energy physics.
🔮 Future Outlook & Predictions
The future of electron manipulation promises transformative technologies. Advances in spintronics, which utilizes the electron's spin in addition to its charge, could lead to faster and more energy-efficient computing and memory devices. Research into topological insulators and 2D materials like graphene is uncovering novel electron behaviors that could revolutionize electronics. The development of more efficient solar cells and batteries hinges on optimizing electron transfer processes. Ultimately, a deeper understanding of electron interactions at the quantum level may unlock entirely new paradigms in energy generation, computation, and materials science.
💡 Practical Applications
Electrons are the workhorses of countless practical applications. In electronics, their controlled flow forms the basis of all modern devices, from smartphones to supercomputers. They are essential for lighting, powering everything from incandescent bulbs to energy-efficient LEDs. In medicine, electron beams are used in radiotherapy to treat cancer, and electron microscopy provides high-resolution imaging of biological and material samples. The chemical industry relies on understanding electron transfer for catalysis, synthesis, and the production of materials like polymers. Batteries and fuel cells harness electron flow for energy storage and conversion, powering electric vehicles and portable electronics.
Key Facts
- Category
- science
- Type
- concept